Until somebody figures out a alternative for lithium-ion in rechargeable batteries, research will continue into find out how to cram extra power inside as well as extending their useful lifespan. Two scientists believe they’ve managed to increase the life of such batteries significantly, and all due to an accident within the lab.
In the present day, lithium-ion batteries usually rely on graphite anodes to offer a long lifespan. Rechargeable battery performance declines and ultimately falls off a cliff (becomes unusable) because of these anodes repeatedly expanding and contracting as lithium ions migrate during the cycle of charging and discharge. Lithium compounds build up on the electrodes throughout this process then break off during the expansion and contraction. This exposes the surface of the electrode and over time decomposes it to the point of failure.
A better alternative to utilizing graphite for the anodes would be aluminum, however aluminum expands and contracts too much during each cycle. If scientists could stop that happening, we’d have much better performing batteries.
Dr Li Ju of MIT, USA and Dr Wang Changan of Tsinghua University, China and have been working collectively to stop the oxide coating that forms on the surface of aluminum nanoparticles when it’s exposed to air. Their idea was to soak the nanoparticles in a sulfuric acid and titanium oxysuplphate mix, which would dissolve the aluminum oxide and replace it with titanium oxide.
Achieving the new outer coating required a set time of soaking. The accident occurred when Wang and Li forgot to remove one batch of the nanoparticles from the soaking process. That batch ended up soaking for several hours longer than intended with the end result being the sulfuric acid and titanium oxysulfate mix leaked into the 50nm nano particles and dissolved some of the aluminum inside. What this left was a nano particle with a 4nm outer shell of titanium hydroxide and an inner 30nm “yolk” of aluminum.
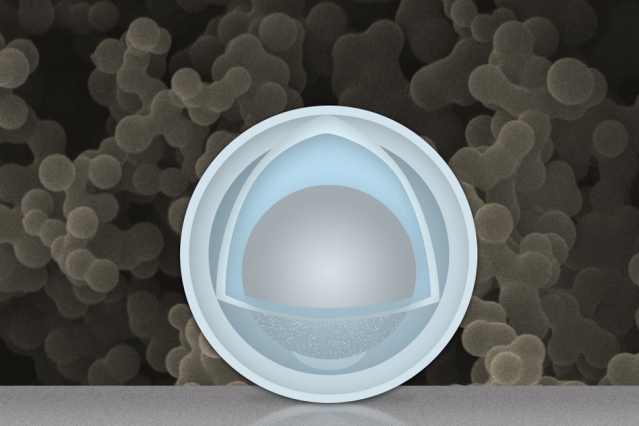
Credits: Christine Daniloff/MIT
Rather than discarding this forgotten batch, they decided to test it by constructing batteries using these particles. It turns out they’ve potentially solved the problem of using aluminum for the anodes in the battery. The extra long soak meant the anodes didn’t expand and contract, in fact they created a battery that over 500 charge/discharge cycles retained as much as 4-times the capacity of the equivalent graphite anode batteries. These batteries last significantly longer in terms of usable lifespan and, according to MIT, can hold up to three-times the energy.
Clearly, sometimes being a forgetful scientist can lead to a breakthrough. The discovery is expected to be an easy one to scale up to mass manufacturing, which means the next-generation of rechargeable batteries may use aluminum anodes, bringing with it longer battery life per charge and batteries which have a much improved lifespan.
“These yolk-shell particles show very impressive performance in lab-scale testing,” says David Lou, an associate professor of chemical and biomolecular engineering at Nanyang Technological University in Singapore, who was not involved in this work.
“To me, the most attractive point of this work is that the process appears simple and scalable.”
The research team included Sa Li, Yu Cheng Zhao, and Chang An Wang of Tsinghua University in Beijing and Junjie Niu, Kangpyo So, and Chao Wang of MIT. The work was supported by the National Science Foundation & the National Natural Science Foundation of China.












